THE IVAN HUC GROUP
Our group has developed a unique know-how in the design, synthesis and characterization of artificial folded molecular architectures (foldamers) based on aromatic oligoamides. These objects differ notably from peptides or nucleotides, from which they are inspired, and from their analogues, and give access to structural motifs and thus to functions beyond those of biopolymers.
Our main research lines include the development of synthetic and design methodologies towards molecular objects as large and complex as small proteins ; the investigation of receptors and molecular motors based on these folded architectures ; the study of potential biological applications of aromatic oligoamide foldamers, in particular to recognize protein surfaces ; and exploration of properties such as electron transport which may be of use in new materials. These projects have fostered a very active collaboration with x-ray crystallographers and our involvement in the set-up of an x-ray diffraction facility.

The group of Prof. Ivan Huc settled at the Department of Pharmacy, in the Faculty of Chemistry and Pharmacy of the Ludwig-Maximilians-University of Munich on September 1th 2017. It is affiliated to the Center for Integrated Protein Science. Strong collaborations and joint PhD supervisions have been established with its former host in France, the CBMN Laboratory, a joint research unit of the CNRS, the University of Bordeaux, the Bordeaux Polytechnic Institute, and with the European Institute of Chemistry and Biology, a research team incubator that promotes emergence and interdisciplinary research on the University of Bordeaux campus. The main collaborations involve the group of Dr. G. Guichard and the group of Dr. Y. Ferrand, both foldamer specialists in CBMN.
News
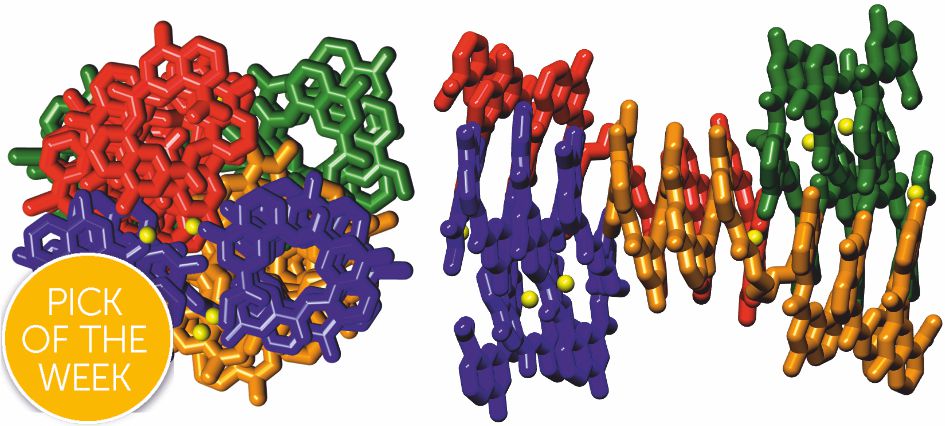
Our eight-helix bundle paper was selected as "pick of the week" by Chem. Sci. editors and the object of a highlight in Chemistry World
Read more
Cover picture: Angewandte Chemie, 2021, 60, 18461 –18466
Read moreHighlight of J. Atcher's paper: "Very Important Publication" in Angewandte Chemie has been highlighted in ChemViews Magazine"
Read more See all newsLatest publications
M. Loos, F. Xu, P. K. Mandal, T. Chakrabortty, C. Douat, D. B. Konrad, M. Cabbar, J. Singer, V. Corvaglia, T. Carell, I. Huc*
D. Bernier, N. Grafl, J. Gnauck , H. Betat, S. Dengler, I. Huc, M. Mörl
L. Wang, C. Douat, J. Sigl, P. Sai Reddy, L. Fischer, B. Langlois Estaintot, Z. Liu, V. Pophristic, Y. Yang, Y. Zhang, I. Huc *



